When Checklists Become Gatekeepers in Reporting Guidelines in Medical Research
Medical research rarely collapses because of bad intentions. It collapses because of weak reporting, selective transparency, and editorial shortcuts. To prevent that, the scientific community created reporting guidelines in medical research—most prominently CONSORT, PRISMA, and STROBE.
These frameworks were meant to clarify science. Today, they increasingly control it.
This article takes a hard, editorial look at how reporting checklists evolved from quality safeguards into informal gatekeepers—and what that means for academic integrity.
Why Reporting Guidelines Exist in the First Place
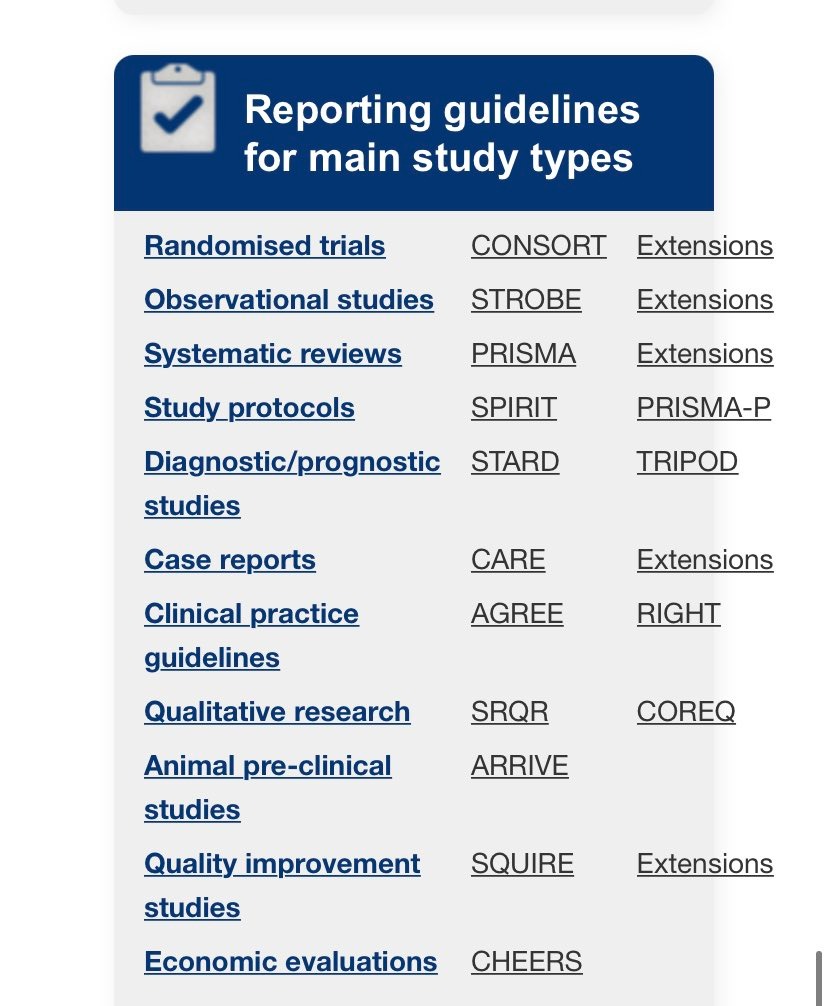
Before standardized reporting, peer reviewers were forced to guess. Methods were vague, outcomes were selectively reported, and replication was nearly impossible. The result was a credibility crisis.
Reporting guidelines in medical research emerged to:
- Standardize disclosure across study designs
- Reduce outcome switching and selective reporting
- Enable reliable synthesis of evidence
The EQUATOR Network centralized this effort by cataloguing validated guidelines and promoting responsible reporting rather than cosmetic compliance.
The intention was guidance. The unintended consequence was power.
CONSORT: From Trial Transparency to Editorial Threshold
CONSORT governs randomized controlled trials and demands explicit reporting of randomization, allocation concealment, blinding, outcomes, and harms.
Scientifically, this is justified. Editorially, it has become restrictive.
Many journals now treat CONSORT like a starter checklist—a paper either “passes” or never reaches peer review. This shifts evaluation away from scientific reasoning toward formatting discipline.
The outcome is predictable:
- Authors learn to write for CONSORT, not from their data
- Context-specific trials are penalized for structural deviations
- Compliance becomes performative rather than informative
CONSORT improves reporting quality, but it does not validate study design. Treating it as a gatekeeper confuses transparency with rigor.
PRISMA: The Authority of the Diagram
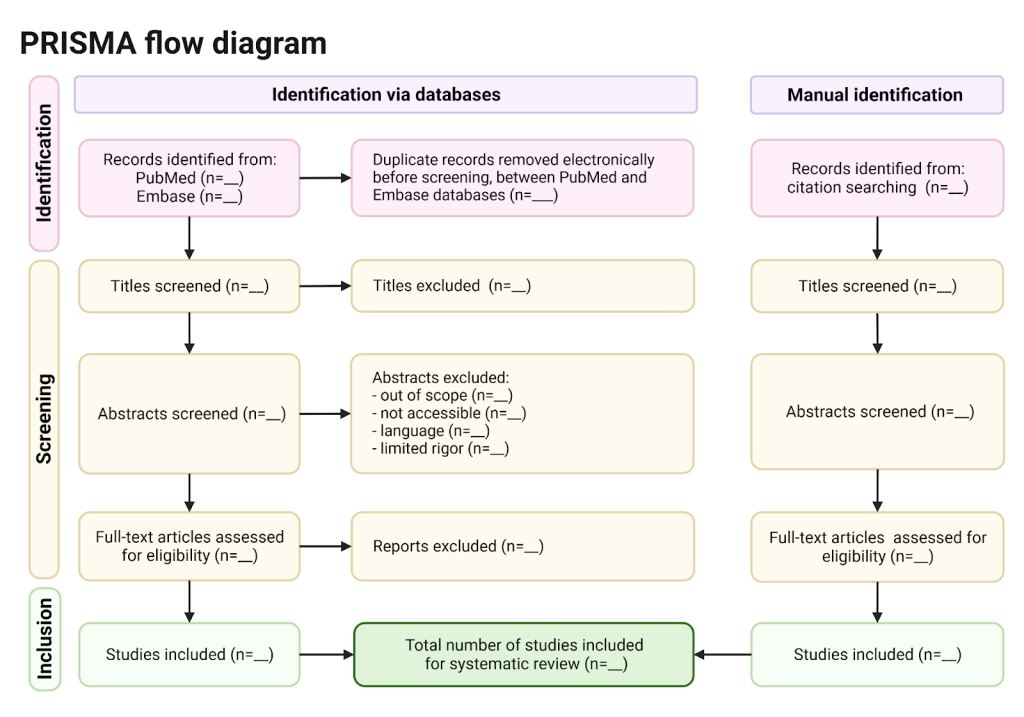
PRISMA dominates systematic reviews and meta-analyses. Its defining feature, the PRISMA diagram, tracks study identification, screening, eligibility, and inclusion.
The diagram is useful. The worship of it is not.
Too often, reviewers assume that a clean PRISMA flow guarantees a strong review. It does not. PRISMA documents process, not judgment.
Weak reviews still pass when:
- Search strategies are biased
- Data extraction lacks reproducibility
- Statistical synthesis is poorly justified
The World Health Organization explicitly stresses that transparent reporting must be paired with critical appraisal, not replace it.
PRISMA is a lens, not a seal of approval.
STROBE: Observational Studies Under Suspicion
STROBE applies to observational research—cohort, case-control, and cross-sectional studies. These designs already face skepticism due to confounding and bias.
STROBE forces authors to disclose:
- Participant selection criteria
- Variable definitions and measurements
- Bias mitigation strategies
Its role is disclosure, not defense.
Yet in practice, STROBE has become symbolic. Some reviewers equate observational research with lower credibility regardless of methodological strength.
As even Wikipedia notes, STROBE improves reporting clarity—it does not certify validity.
The checklist cannot compensate for poor reasoning, nor should it disqualify sound observational science.
When Checklists Start Acting Like Gatekeepers
Here is where reporting guidelines in medical research cross a dangerous line.
Instead of informing review, checklists increasingly:
- Drive desk rejections
- Replace substantive peer critique
- Encourage template-driven manuscripts
This mirrors checklist behavior outside academia. A hospital bag checklist ensures nothing is forgotten—but it does not guarantee a safe delivery. Even a hospital bag checklist UK prepares for logistics, not outcomes.
In publishing, checklists now function like a strobe light—highly visible, impossible to ignore, and sometimes blinding to nuance.
CONSORT vs PRISMA vs STROBE: What They Do—and What They Don’t
| Guideline | Study Type | What It Improves | What It Cannot Do |
| CONSORT | Randomized controlled trials | Transparency of trial conduct and outcomes | Fix flawed randomization or poor ethics |
| PRISMA | Systematic reviews & meta-analyses | Traceability of study selection | Guarantee unbiased synthesis |
| STROBE | Observational studies | Clarity of methods and variables | Eliminate confounding or bias |
This distinction matters. Reporting guidelines in medical research are disclosure tools—not quality certificates.
Editorial Convenience vs Scientific Responsibility
Journals rely on checklists because they appear objective. A checklist feels safer than judgment.
But outsourcing evaluation to frameworks does not eliminate bias—it disguises it.
Even major science publishers have begun questioning this dependency. Nature has warned against checklist absolutism, emphasizing that editorial responsibility cannot be automated.
The checklist should support editorial judgment, not replace it.
Where ClinicaPress Stands
At ClinicaPress, reporting guidelines are treated as instruments, not verdicts.
Our editorial philosophy prioritizes:
- Context-aware guideline use
- Methodological reasoning over mechanical compliance
- Transparency without performative formatting
We routinely encounter manuscripts weakened by checklist anxiety and others artificially strengthened by cosmetic adherence. Both distort the scientific record.
How Authors Should Use Reporting Guidelines Without Losing Integrity
Responsible use of reporting guidelines in medical research follows a simple rule: design first, disclose second.
Best practice means:
- Using guidelines during study planning, not post hoc
- Explaining justified deviations transparently
- Writing for human readers, not automated filters
Our recommendations for navigating reviewer expectations without compromising integrity are detailed in our guide on Peer-Reviewed Journal Explained: 10 Reasons Why It Matters for Academic Research and expanded through our editorial policies.
The Way Forward: Guidance, Not Gatekeeping
The solution is not abandoning CONSORT, PRISMA, or STROBE. It is restoring their original purpose.
Reporting guidelines in medical research should:
- Illuminate decision-making
- Expose limitations honestly
- Support—not replace—scientific judgment
Science advances through clarity, not compliance theater. And checklists should never decide who gets to speak—only how clearly they do.